HRPP Toolkit updates and VIRBs Tips
| *HRPP Toolkit updates* |
| ATTN: VCU Research Communities The VCU HRPP completed the migration to VIRBs on 8/7/2025. As part of this process, all toolkit documents were updated. Below is a summary of the changes made. All HRPP Toolkit documents including, SOP’s, General Documents and Forms, Templates, IRB Reliance Forms, Worksheets, and Checklists have been updated to align with VIRBs and updated review process, and revised branding. The current versions of the HRPP Toolkit materials are available in the VIRBs Library feature and are also available in the HRPP Toolkit. ***Check out our tips below for more information on the VIRBs library feature!*** |
| Look out for the following 8/7/25 updates in the HRP-503 protocol and HRP-502 consent templates: HRP-503 Template Protocols The HRP-503 – TEMPLATE PROTOCOL has been expanded to include additional guidance and instructions for addressing HIPAA applicability and criteria for requests of HIPAA waivers, use of Veeva for e-consent, additional tables included at the beginning of the template (Study Summary section) to capture items that were in the previous smartform, corrections to formatting and editorial changes for ease of use. The HRP-503a – TEMPLATE SBS PROTOCOL has been expanded to include additional guidance and instructions for addressing HIPAA applicability and criteria for requests of HIPAA waivers, additional tables included at the beginning of the template (Study Summary section) to capture items that were in the previous smartform, corrections to formatting and editorial changes for ease of use. The HRP-503b – TEMPLATE NHSR has been expanded to address submissions of QI/QA projects with VCU HS PROC determinations, revised Section 5.0 Data and/or Specimen Collection, and revised 6.0 HIPAA section re DUA for LDS from VCUHS, corrections to formatting and editorial changes for ease of use. Updated versions of the HRP-503 protocol templates are available in the VIRBs Library feature and are also available in the HRPP Toolkit under ‘Templates’. HRP-502 Template Consent The HRP-502 Consent template has been updated to address NIH-funded research involving digital health technologies. Reliance Stack/Forms (800 Series) The Reliance toolkit documents were refreshed to better detail the reliance process at VCU. The Reliance forms are generally referred to as the ‘800’ series. Reliance/external Toolkit materials have been uploaded into the VIRBs Library feature and are also available in the HRPP Toolkit under ‘IRB reliance forms’. HRP-103 – Investigator manual and HRP-103p – Investigator manual – Single IRB review of multi-site research (pSite) The HRP-103/HRP-103p was updated to clarify what is required when investigators rely on an external IRB and when an investigator adds a study site to protocol reviewed by the VCU IRB. Additionally the following items were clarified: – VIRBs process requirements – Requirements for using the short form consent – Process for changing PI and how to handle unanticipated PI leave – GCP, DoD, DoE and VA Requirements (Appendices) Other toolkit documents were updated to reflect the HRPP processes. |
| *VIRBs Tips* |
| Ancillary Reviews (Current status for AR functionality; reminder error – awareness only; department reviews) VIRBs has a built-in feature for completing required Ancillary Reviews within the history of the VIRBs protocol. Currently, the Departmental Review requirement for Expedited and Full Board Studies is the only Ancillary Review that may be completed in VIRBs. The HRPP will notify the research community when any new features become available in VIRBs. Refer to the New IRB review system: VIRBs (VCU IRB system) web page under ‘Ancillary review process‘ for guidance on Department Head Reviews. The Department Head Ancillary Review in VIRBs video demo is available on the HRPP Kaltura channel. To note, VIRBs does not automatically prompt the research team to complete Departmental Review for Expedited and Full Board studies. This is a change from the previous review system. As part of the VIRBs intake process, the IRB immediately returns initial Expedited and Full Board submissions to the research team if Departmental Review is not complete. *Known Error* Ancillary Review Reminders The HRPP is aware of reminder notifications being prompted in the history of VIRBs submissions. Research teams only need to complete the Ancillary Review activity in VIRBs for initial review of Expedited and Full Board studies, or when required to do so as part of the IRB’s review process for a specific protocol. For all other studies, please disregard the automatic notification for ‘VCU Ancillary Review Reminder’ in VIRBs. Location for approved (date stamped) consent forms Make sure you check the ‘Documents’ tab in the submission for the finalized, stamped version, as that version does not auto-populate in the SmartForm. 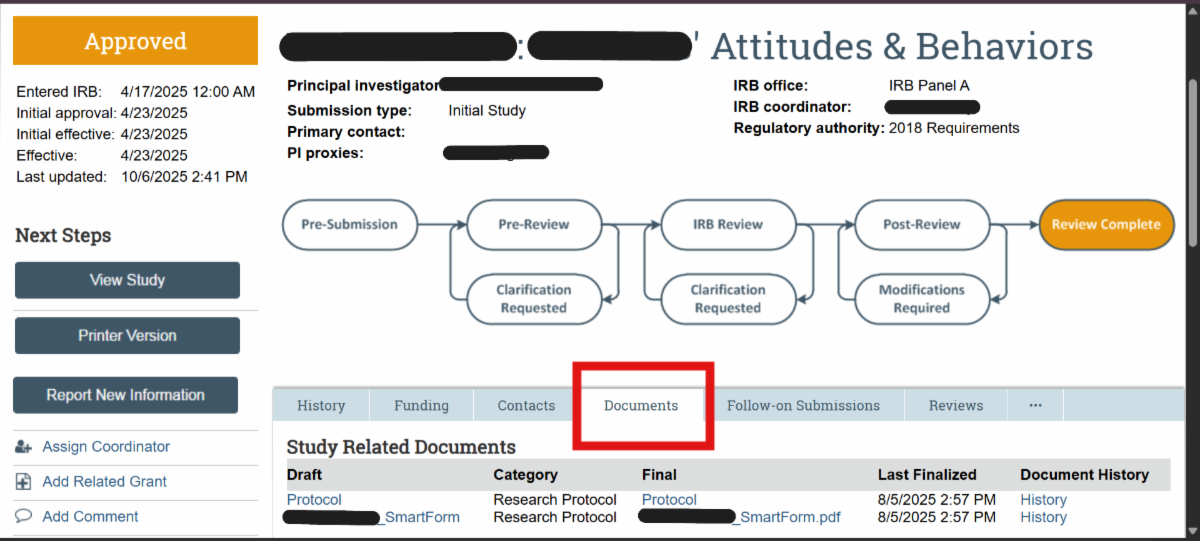 If you have a Modification to the Consent form that is in progress and need to retrieve the currently approved (stamped) version of the consent form, you will need to navigate to the ‘title’ of the study in the breadcrumbs and navigate to the ‘Documents’ tab that contains all currently approved documents. 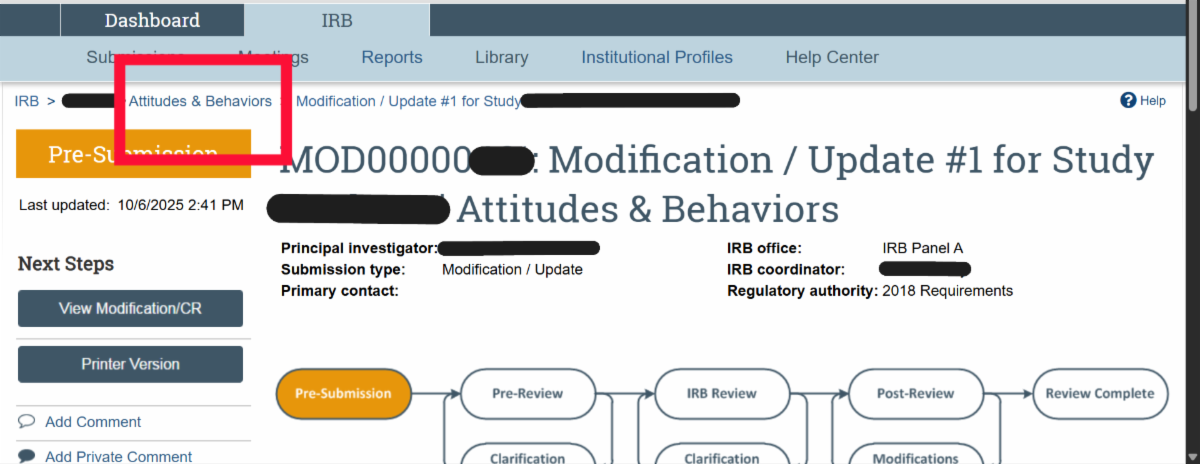 VIRBs Library The VIRBs library is a feature built into VIRBs for direct retrieval of the most up to date versions of the HRPP Toolkit materials, such as SOP’s, protocol and consent templates, and other supporting materials for the IRB’s review process. The VIRBs library will be updated when new versions become available. The HRPP also publishes updated HRPP Toolkit materials on the HRPP Policies and Guidance web page. 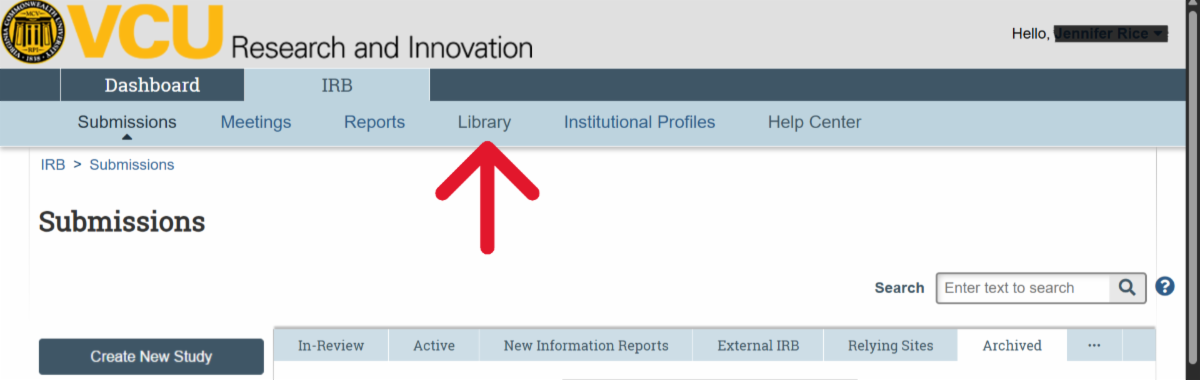 VIRBs Notifications VIRBs is not designed to send auto-notifications for every activity completed. VIRBs includes a built-in flow diagram for tracking of the submission through the IRB’s review process. VIRBs sends auto notifications when comments are logged, when clarifications are needed, and for studies that have been approved by the VCU IRB. For reliance studies receiving review by an external IRB, the VIRBs review process is abbreviated, as it is specific to the processing of external submissions and formal institutional agreements for collaborating studies. VIRBs does not currently send notifications out when external submissions are complete. The HRPP Reliance team has been logging comments to the research teams once external reviews have been completed in VIRBs. |
| *General VIRBs Guidance* |
| VIRBs demos and training videos can be found on the New IRB Review System web page and on the HRPP Kaltura Channel. To see our progress related to VIRBs implementation, refer to the ‘VIRBs transition in process’ drop down found on the New IRB Review System web page. For all important upcoming dates and deadlines and to learn more about transitioning to a protocol-based application the HRPP website visit New IRB Review System: VIRBs (VCU IRB Review System). Reach out to [email protected] with questions about individual study requirements. ___________________________________ |